Regulatory work,
made faster
Cut hours from tedious QA/RA tasks,
with AI tools you can actually trust.
Trusted by 500+ customers
OUR VISION
Less manual work,
more meaningful work
You're under pressure to move fast, but cutting corners isn't an option. Every document needs to be right, every source traceable, but you're spending hours on tasks that can feel frustratingly manual.
Your expertise is what gets devices to market safely.
It shouldn't be wasted on busywork.
OUR FEATURES
Move faster
without cutting corners
Each feature is designed to save you time
and ensure compliance with the latest regulations.

Regulatory Watch
Stop drowning in regulatory updates
Intelligence on what changed in your regulatory landscape filtered for your device type and markets. Know exactly what changed and what you need to do.
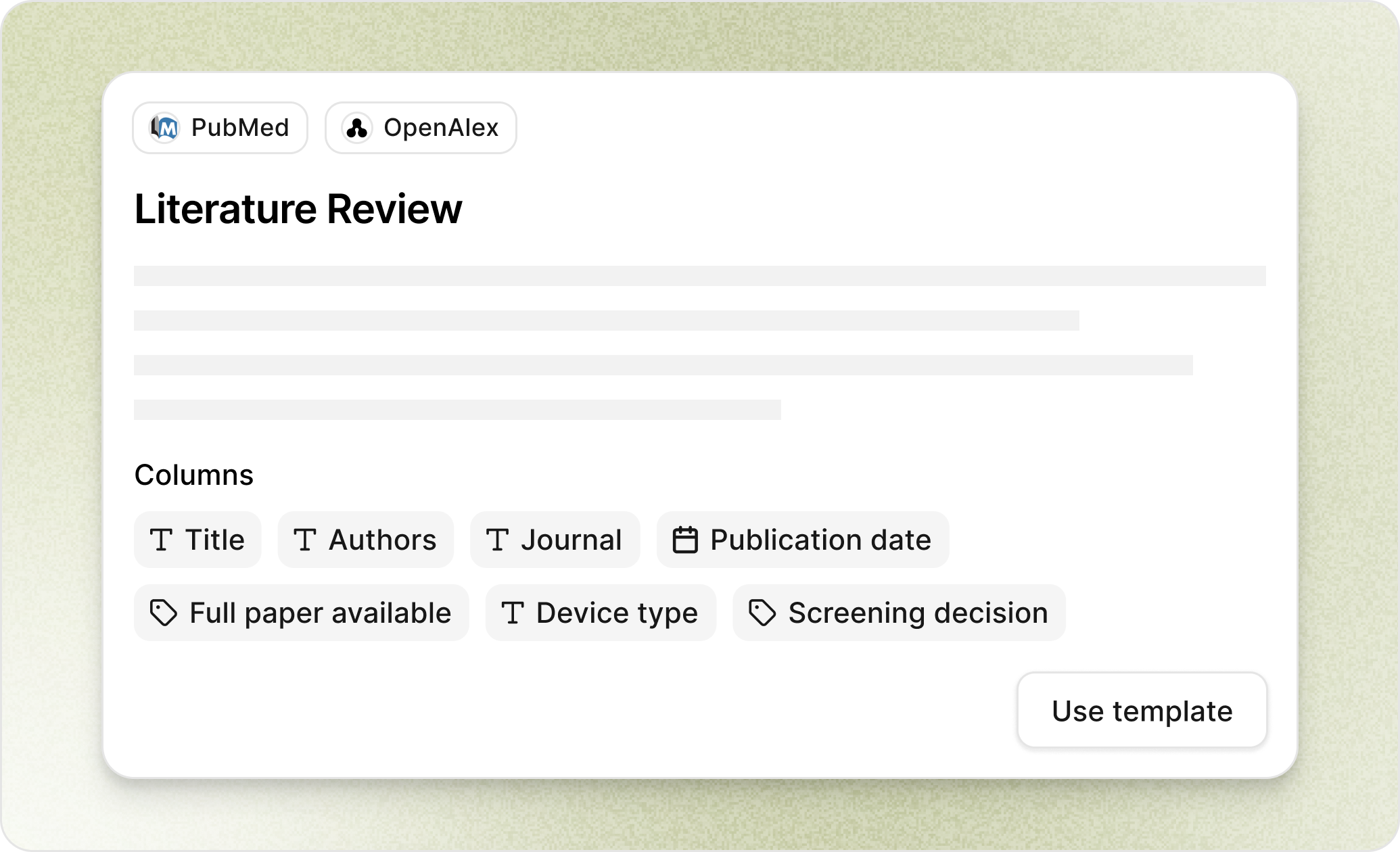
Literature Review
Automate your systematic literature reviews
We search PubMed and scientific databases, extract relevant data, and format results with every source cited and auditable.
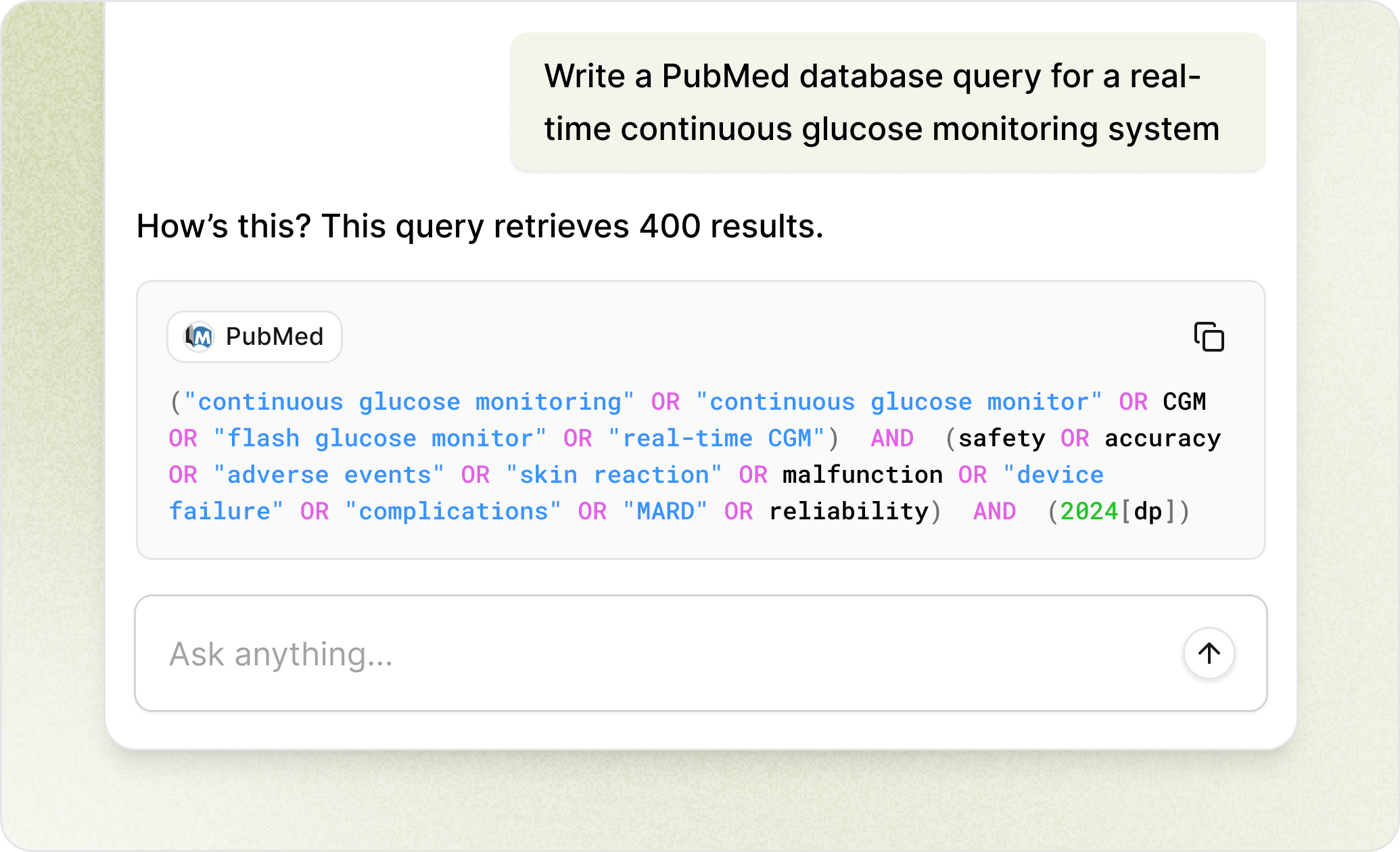
Expert Chat
Instant regulatory answers, always cited
Ask complex QA/RA questions, get answers grounded in regulations and standards, with sources you can verify.
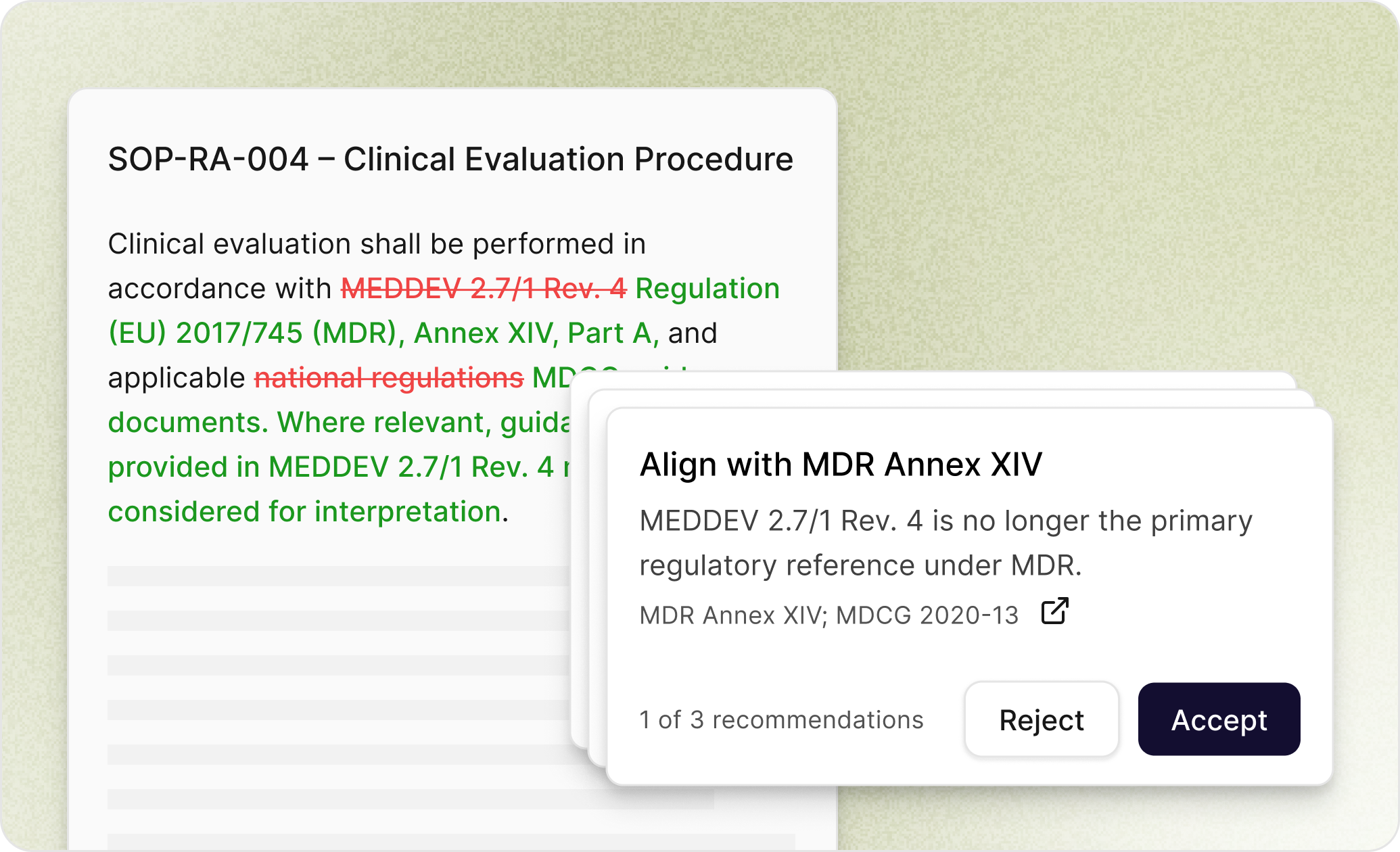
Augmented QMS
No more manual chasing. Focus on what matters.
Connect your stack and let AI handle the vigilance: sizing changes, tracking impact, catching gaps, and moving requests forward with full traceability and control.
Want a complete platform tour?
Request a demoCOMPREHENSIVE SOURCES
All the official sources,
all in one place
Trained on millions of pages of regulatory documentation. All sources are regularly updated and verified by QARA experts.









European Commission, FDA, MDCG/MEDDEV, ISO/IEC Standards, IMDRF, US Federal Register, EU NANDO, National Authorities, PubMed, MAUDE, EMA, EFSA, and more...
HOW IT WORKS
Tailored to your device, your markets, your needs
Generic tools give generic answers. Qalico learns about your specific situation to deliver intelligence that actually matters.
01
Set up your profile
Tell us about your device, product codes, target markets, and clinical claims.
02
We filter for relevance
Our AI uses your profile to search, monitor, and analyze only what applies to you.
03
Get personalized results
No noise — just cited answers, regulatory updates, incident alerts, and documentation tailored to your context.
Ready to simplify your regulatory work?
Join QARA professionals already using Qalico to save hours, weekly.