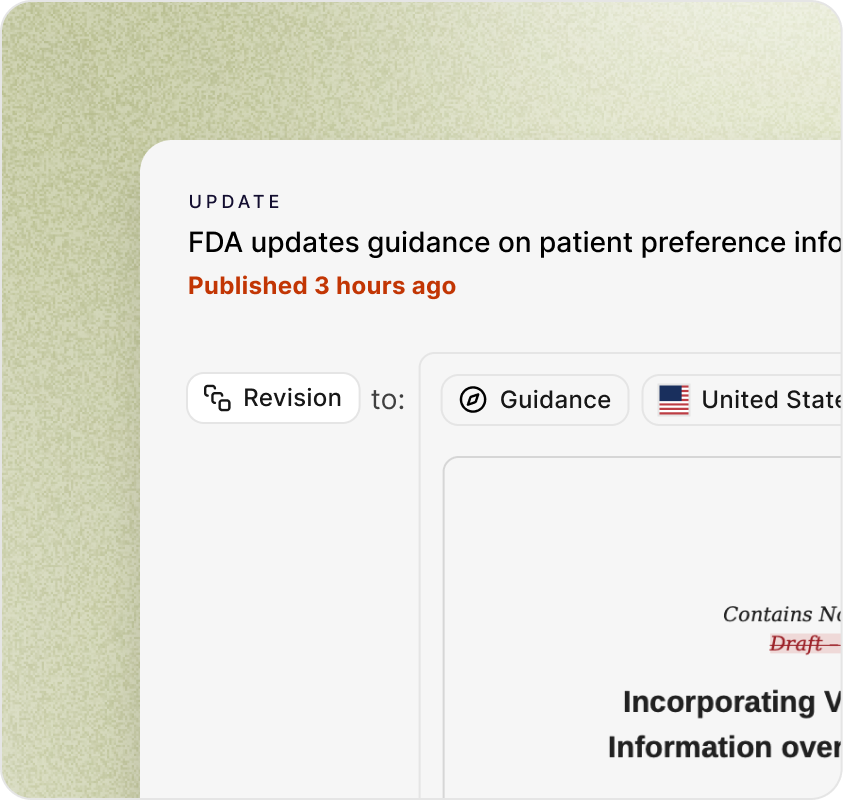
Real-time change detection
Every page is fingerprinted. When a document, guidance, or standard is updated, Qalico detects the diff within hours, not weeks.
Qalico watches every authority, standards body, and notified body your devices depend on — then classifies and routes each update to the right product, reviewer, and process.

Authoritative coverage, globally: Over 200 regulatory bodies, standards organizations, and notified bodies monitored continuously — FDA, EMA, MHRA, Health Canada, PMDA, NMPA, ISO, IEC, IMDRF, and dozens more.










how it works
The bottleneck in regulatory monitoring isn't access to documents; it's filtering signal from noise. Qalico crawls 200+ authoritative sources on a fixed cadence, captures every new publication with a full audit trail, scores each update against your registered portfolio, and routes the relevant ones to the people who own the response. Reviewers stop scanning weekly digests and start receiving targeted, actionable items.

Every page is fingerprinted. When a document, guidance, or standard is updated, Qalico detects the diff within hours, not weeks.
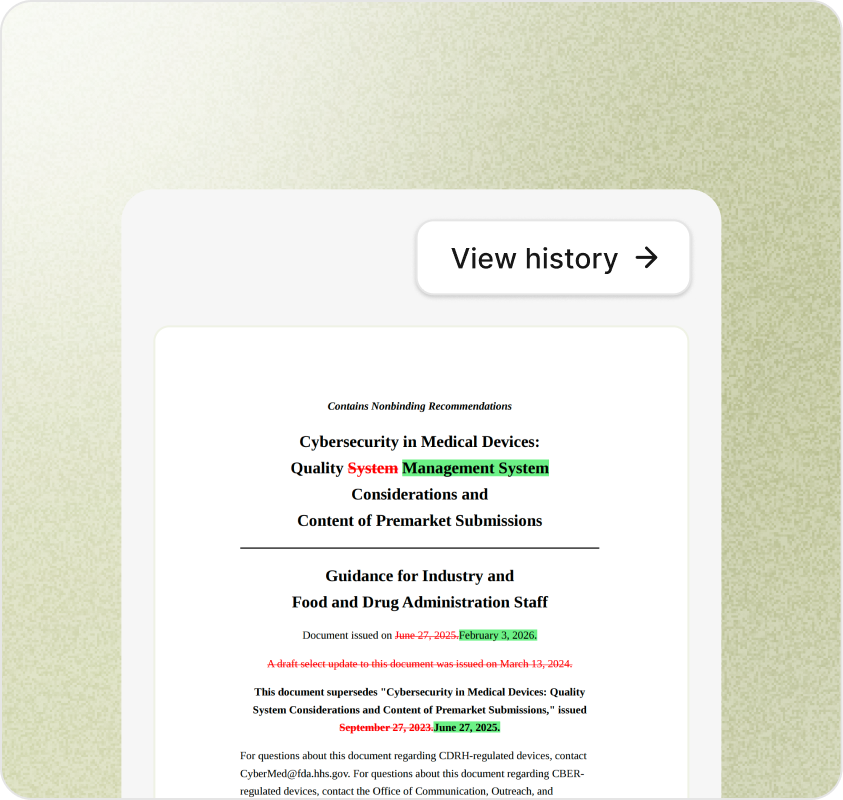
Each captured update is stored with its original URL, retrieval timestamp, and source hash — so you can prove what was published and when, even after agencies revise or delete it.

Updates are scored against your registered device portfolio, product codes, intended uses, classifications, and target markets, so reviewers only see what applies.
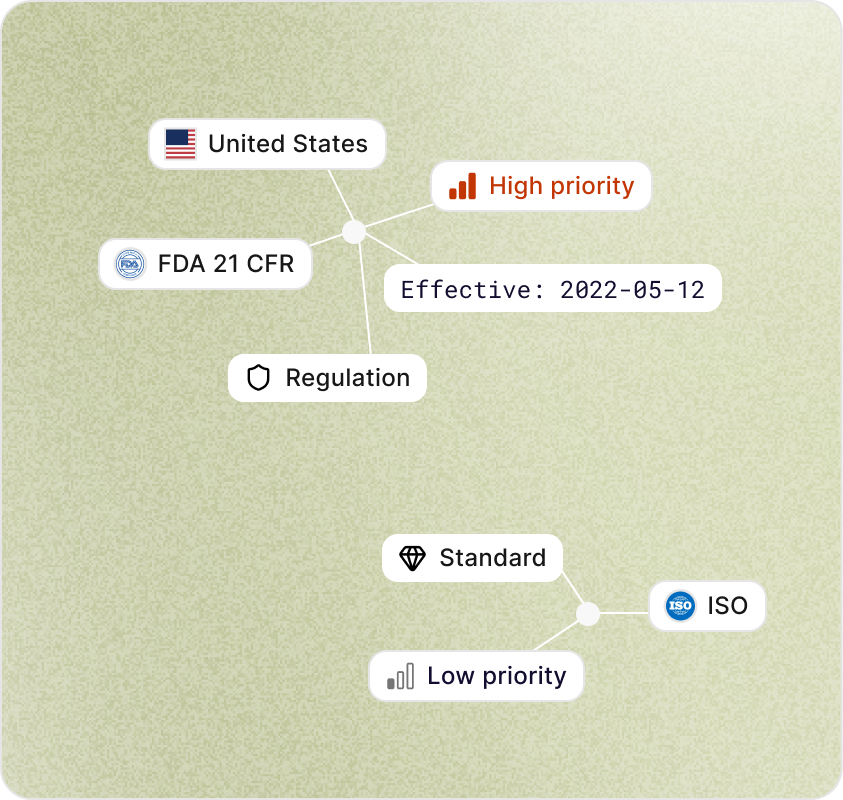
Each update is tagged across jurisdiction, document type, topic (cybersecurity, biocompatibility, software, etc.), affected device classes, and effective dates.

High-impact updates (new obligations, imminent effective dates, recalls) are flagged automatically so they jump the queue.

Native-language documents from non-English jurisdictions are captured and translated, so coverage isn't limited to what publishes in English.
Book a 30-minute walkthrough tailored to your device portfolio and regulatory scope.