Post-Market Surveillance
AI-enabled safety database monitoring. Automate adverse event tracking and stay ahead of regulatory signals.
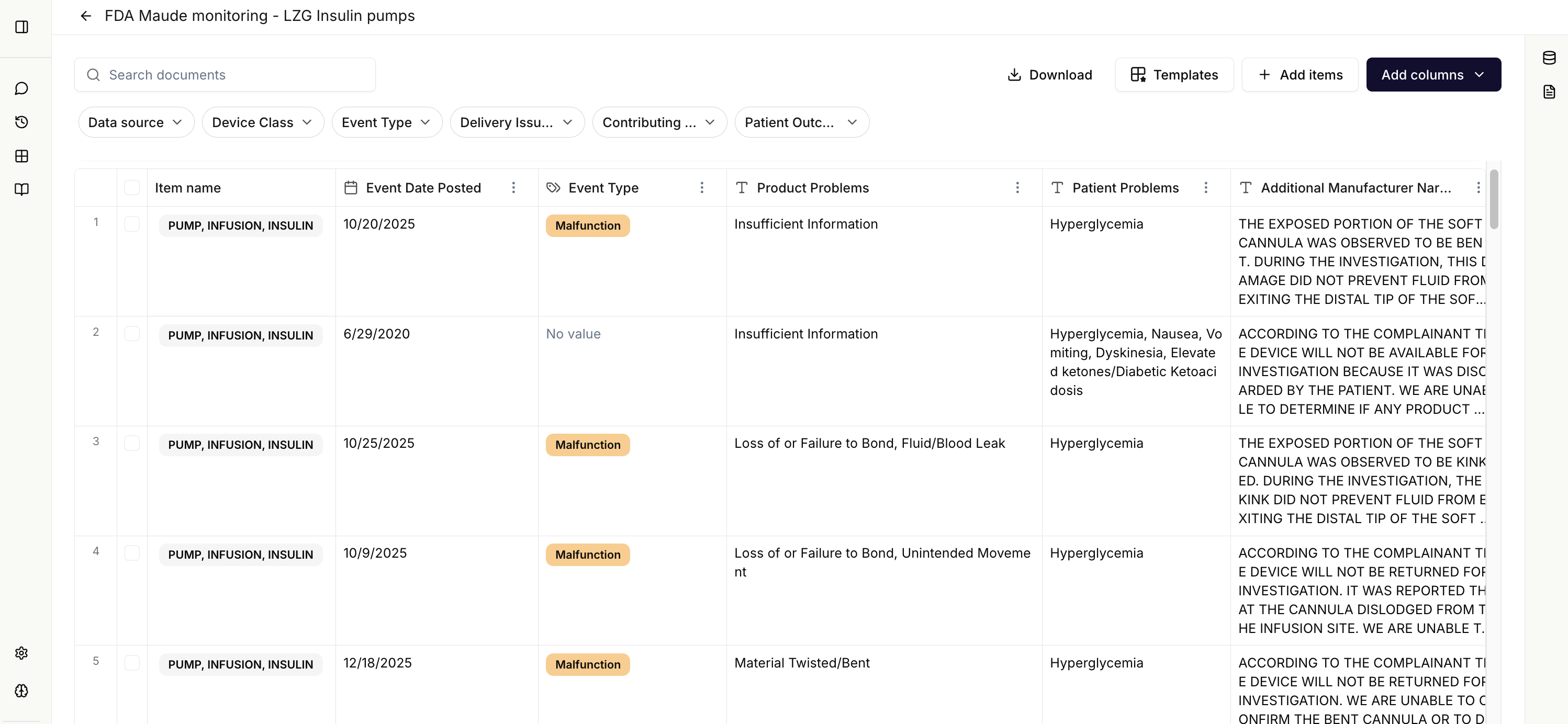
FDA MAUDE & Recall Data Aggregation
Automatically aggregate and structure data from FDA MAUDE adverse event reports and FDA recall databases in one unified view.
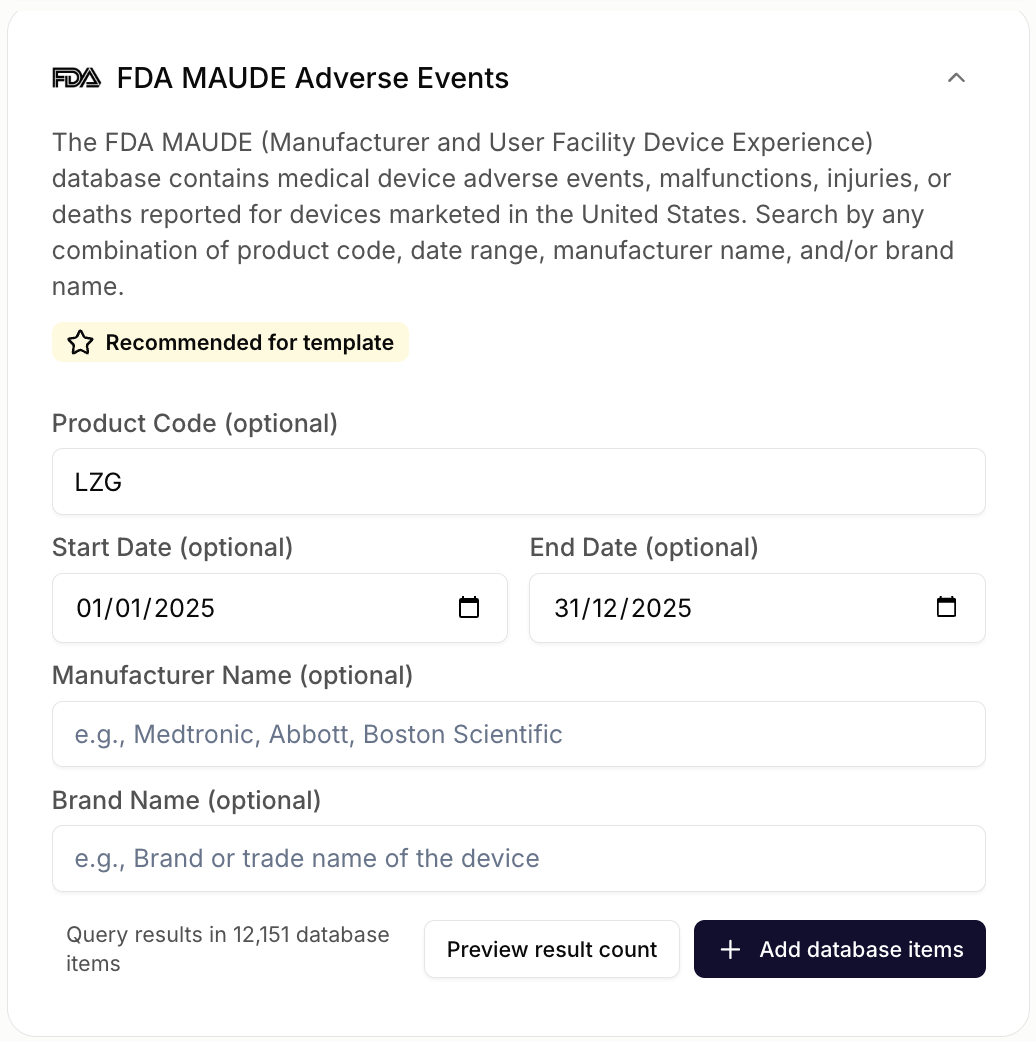

AI-Enabled Data Extraction at Scale
Let AI extract patterns, identify trends, and surface insights from large volumes of safety data. Analyze thousands of reports in minutes.
Grounded in Truth
Every insight is traceable back to its source. AI-generated analysis comes with full justification and citations, so you can verify and trust the results.

Interested in Post-Market Surveillance?
Talk to one of our experts.
Book a demo