The UK's MHRA just published the most comprehensive refresh of clinical investigation guidance since Brexit.
Between March 30 and April 1, the MHRA released guidance covering clinical investigations in Great Britain, Northern Ireland, submission requirements, approval processes, biological safety assessments, statistical considerations, investigators' responsibilities, and electrically powered devices.
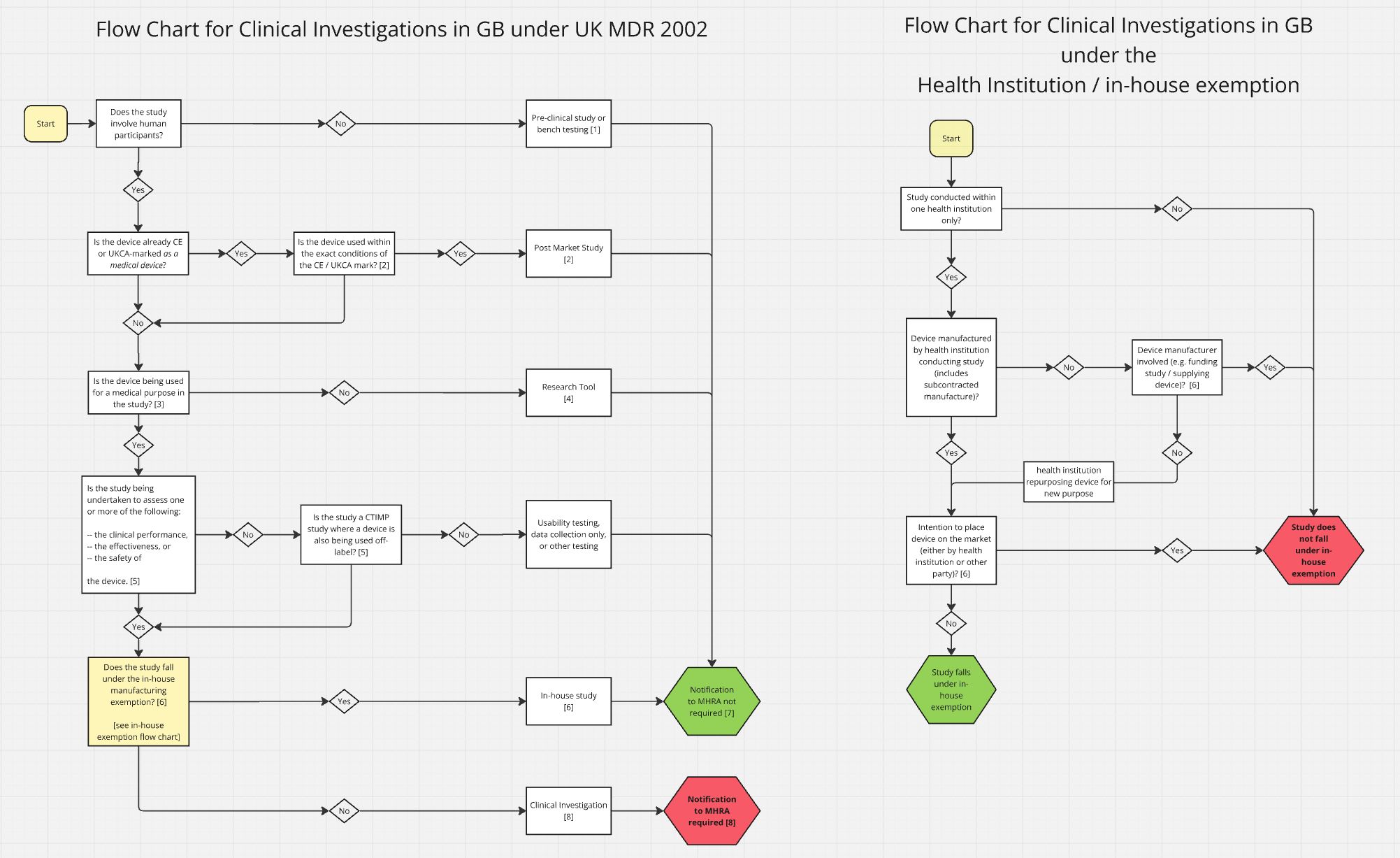
MHRA put together two flowcharts to determine whether you will need to undertake a clinical investigation.
The main clinical investigation pathway
The first chart walks through the main clinical investigation pathway under UK MDR 2002: does your study involve human participants? Is the device already CE/UKCA-marked and used within the exact conditions of that marking? Is it being used for a medical purpose? Depending on the answers, you land on a post-market study, a research tool study, an in-house study (no MHRA notification required), or a full clinical investigation.
The health institution and in-house exemption
The second chart covers the health institution and in-house exemption: is the study conducted within one institution only? Is the manufacturer involved? Is there intent to place the device on the market? This determines whether the in-house exemption applies or not.
If you are running or planning device studies in the UK, these documents should be on your reading list.
Qalico has indexed the full set so our users can track these alongside their existing regulatory landscape.