Latest from Etienne

We interviewed 100+ QARA professionals about AI. Here's what we learned.
Over the past year, we talked to over 100 regulatory and quality professionals about AI. We turned those conversations into a practical guide.

FDA Draft Guidance: Reduced Oversight for NIOSH-Approved Respirators
FDA proposes stepping back from duplicating NIOSH oversight for approved respirators in healthcare settings. Here is what matters for manufacturers.

MHRA AI Airlock: The World's First AI Medical Device Regulatory Sandbox Gets a 3-Year Extension
The MHRA extends its AI Airlock programme with 3 years and £3.6 million in funding. Here is what two phases of testing have revealed for manufacturers.

IMDRF Opens Consultation on AI Medical Device Lifecycle Framework
The IMDRF's new AI framework (N93) will shape the global regulatory baseline for AI-enabled medical devices. Here are the four most critical takeaways for manufacturers.

FDA Finalizes Patient Preference Information Guidance: What Changed From Draft to Final
FDA finalized its guidance on incorporating voluntary Patient Preference Information over the total product life cycle. We redlined the changes from the 2024 draft.
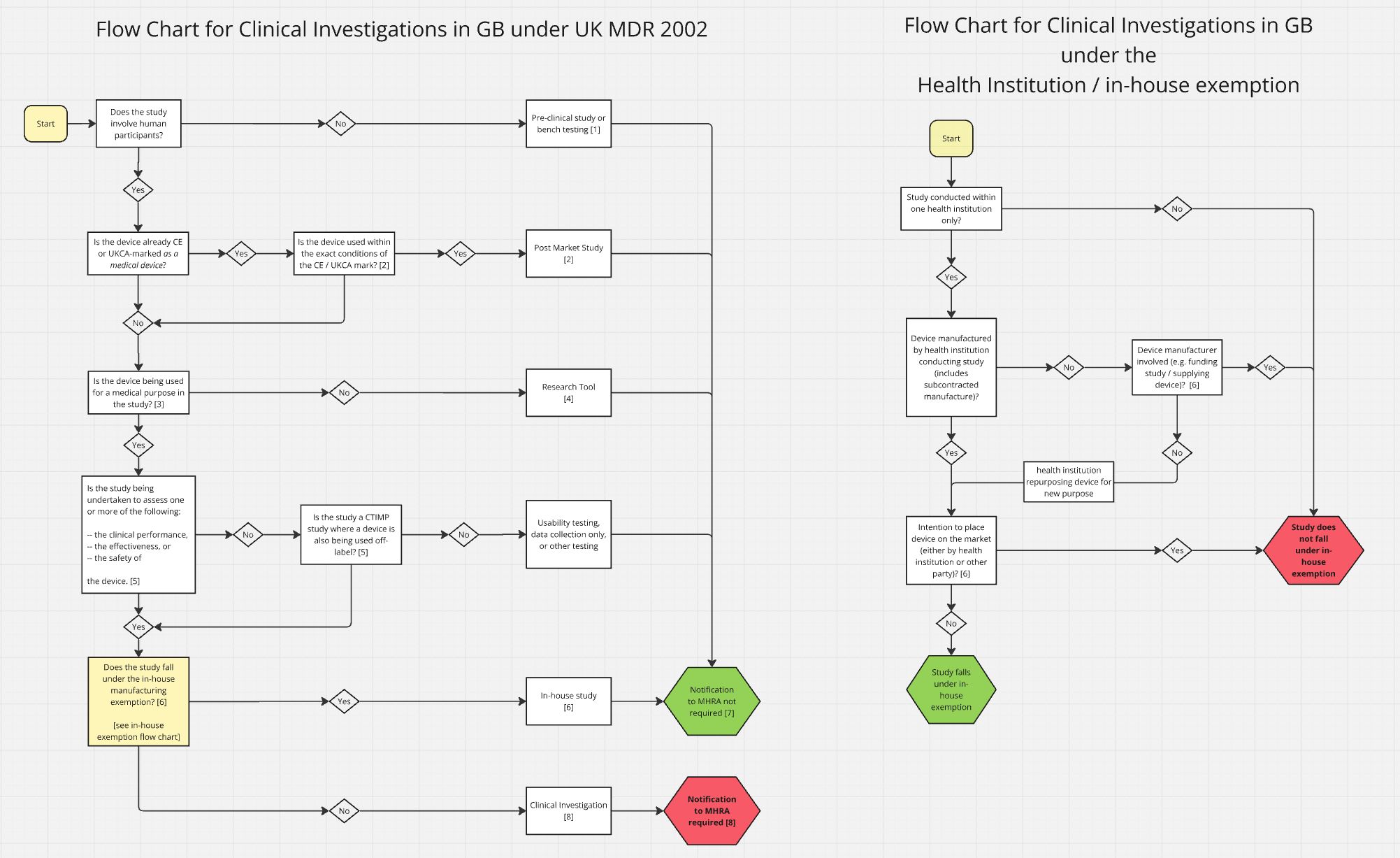
MHRA Publishes Most Comprehensive Clinical Investigation Guidance Since Brexit
Between March 30 and April 1, the MHRA released guidance covering clinical investigations, submission requirements, approval processes, and more.

When NOT to Use AI in Your Regulatory Work
Generalist AI keeps improving, but it is still far from enough for proper regulatory work. Here are five scenarios where you should not rely on it.

From Guidance Document to Requirements Checklist in 2 Minutes
A reusable prompt to extract every requirement from any regulatory guidance into a structured, traceable table. Applied here to the FDA Clinical Decision Support Software guidance.
